BiQ: Update on Darlifarnib from Kura Oncology's IKCS Investor Event 04/17/26
Kura Oncology (KURA) hosted an investor event today to discuss updated data from its ongoing Phase 1a/b dose-escalation trial for Darlifarnib in combination with Cabozantinib for the treatment of ccRCC. In this article, I don't plan to reiterate all the information from today's presentation, since it's easily accessible in KURA's presentation materials; instead, I will focus on what I found most interesting about Darlifarnib. Readers can familiarize themselves with today's presentation by visiting the events page on the Kura Oncology website:
https://ir.kuraoncology.com/events/event-details/kura-oncology-ikcs-virtual-investor-event
Readers can also visit my earlier article on KO-2806 (Darlifarnib) here (paywall removed).
The primary cause of 80% of RCC tumors is VHL mutations, which causes the accumulation of HIF-2α in the cell. This, in turn, drives massive, non-stop production of VEGF, which leads to the characteristic hyper-vascularization observed in RCC tumors. TKIs, such as Cabozantinib, suppress the VEGF pathway, which makes them effective for the treatment of RCC. However, a common escape mechanism in RCC tumors is the RHEB/mTORC1 pathway, which allows them to bypass the VEGF pathway altogether. This is what leads to Cabo desensitization; eventually, patients become refractory to Cabo.
Darlifarnib, previously known as KO-2806, is KURA's 2nd-generation Farnesyl Transferase Inhibitor (FTI). What makes Darlifarnib unique is that it very specifically targets RHEB, unlike 1st-generation FTIs, such as Tipifarnib, which target the RAS pathway. Also, Darlifarnib is designed to specifically block the mTORC1 pathway, while sparing mTORC2, by preventing farnesylation of RHEB. There are several key differences that make Darlifarnib potentially superior to 1st-generation FTIs, especially for RCC:
- Previous FTIs were not specific enough to target mTORC1 while sparing mTORC2. This led to significant toxicity, as mTORC2 inhibition is toxic to healthy cells.
- Previous FTIs, such as Tipifarnib, were dosed at up to 600mg twice-daily, as opposed to 3mg or 5mg once-daily for Darlifarnib. This significantly limits off-target toxicity.
- Darlifarnib has a significantly longer half-life, which allows for a pulsed-dosing regimen of 7-days on and 7-days off. This pulsed dosing regimen allows time for healthy cells to recover.
- By targeting the RHEB pathway specifically, Darlifarnib avoids the GGTase escape mechanism that plagues earlier RAS-directed FTIs. This is especially important in RCC, which is a RHEB-driven cancer.
By blocking the RHEB/mTORC1 pathway, which Darlifarnib does very effectively, Darlifarnib closes the "escape hatch". Essentially, while a TKI closes the front door, Darlifarnib blocks the back door. This is the mechanism underlying the combination of Darlifarnib with a TKI, such as Cabo, for the treatment of RCC.
The updated data presented earlier today by KURA appears to lend significant credence to this hypothesis. Darlifarnib + Cabozantinib demonstrated an ORR of 44% and a DCR of 94% in Cabo-refractory patients.
Although these results are based on a relatively small patient population, I find them to offer meaningful proof of concept for the hypothesis. I am looking forward to additional data, which will hopefully also provide some clue as to potential durability.
What's equally as interesting is the safety profile of Darlifarnib. Not only does Darlifarnib avoid additional toxicities (except Neutropenia, which is relatively easy to manage), but it also appears to mitigate many of the AEs associated with Cabo, which is something I also noted in my earlier article:
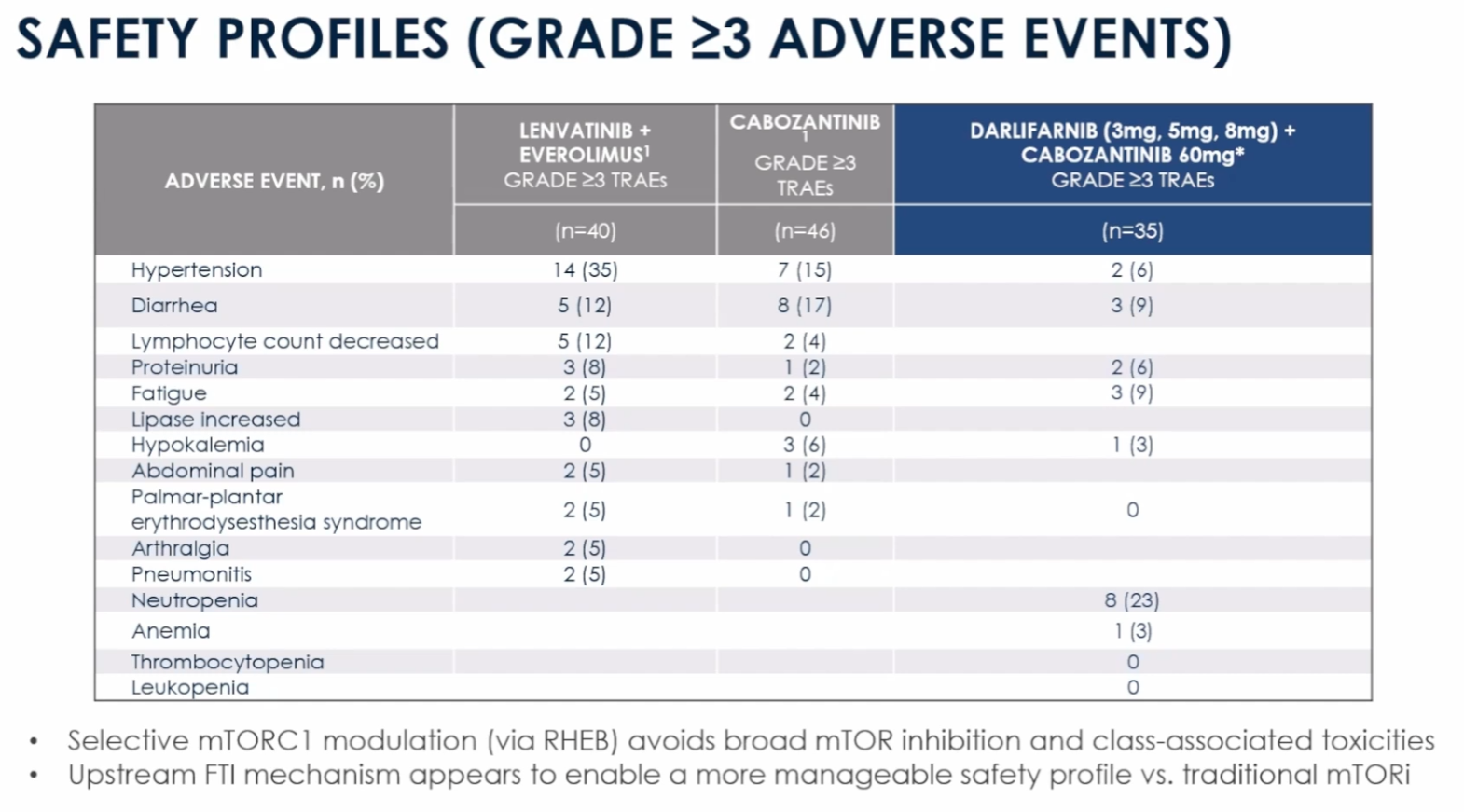
What follows below are my own thoughts; they did not originate from KURA's presentation, so take them fwiw.
How could Darlifarnib + Cabozantinib have a safer tox profile than Cabozantinib alone? While this may seem counterintuitive, I think it may actually make mechanistic sense: by restricting the RHEB/mTORC1 pathway, Darlifarnib prevents healthy cells from overstimulating mTORC1, which can lead to inflammation and fatigue. Over time, hyperactivation of this pathway can cause patients to become intolerant of Cabo, narrowing the therapeutic window and leading to the commonly observed dose reductions. By limiting the damage caused by mTORC1 hyperactivation, Cabo-associated toxicities might be somewhat mitigated. Again, these are my own thoughts, but if correct, this could potentially make Darlifarnib+Cabo even more attractive than Cabo alone for TKI-naive patients.
On today's webcast, management also mentioned plans to trial Darlifarnib in combination with KRAS inhibitors for the treatment of KRAS-driven cancers, such as NSCLC, PDAC, and CRC. Mechanistically, I think this makes a lot of sense. One of the main resistance mechanisms in KRAS-driven cancers is activation of the RHEB/mTORC1 pathway to escape KRAS inhibition, making Darlifarnib potentially ideal in combination with KRAS or pan-KRAS inhibitors. If subsequent trials verify this hypothesis, KRAS-driven tumors could be an even much larger potential market for Darlifarnib than RCC.
One analyst on today's call asked how Darlifarnib may fit into the treatment paradigm for RCC, given the growing importance of HIF-2α inhibitors, which have become an important 2L treatment after the failure of 1L IO therapies. As discussed above, RCC is driven by mVHL, leading to an accumulation of HIF-2α in the cell. By inhibiting HIF-2α, drugs such as Belzutifan and Casdatifan work upstream of VEGF and have demonstrated very encouraging efficacy, especially in combination with a TKI.
Management responded that Darlifarnib would most likely be positioned as a rescue therapy for patients refractory to 2L HIF-2α/TKI combinations. I think this makes sense. However, and once again I'm sharing my own thoughts, given Darlifarnib's complimentary MOA and safety profile, I wouldn't rule out triple combinations in the future. Either way, if future trials are successful, I think Darlifarnib could be an important addition to TKI backbone therapies in RCC, and also potentially used in combination with KRAS-directed therapies in KRAS-driven cancers.
Please refer to the BiQAP Live spreadsheet on the Active Portfolio page or the iQCS for additional information.
KURA share price at time of publication: $9.68
BiQ ACB: $6.11
Please note that the BiQ ACB includes any open options positions, which are accounted for using a "worst case scenario" assumption, plus trading profits (and losses) around my core position.
Not a BiQ Premium member, or have a friend who may be interested? Try BiQ Premium for $50 per month, or $375 (25% off) for the first year.
Biotech iQ is 100% subscriber supported. If you find this information helpful, please spread the word. You can also follow me on X @ Biotech iQ (_Biotech_iQ).
Biotech iQ is not an investment professional, and nothing on this page or this website should be considered investment advice. Please consult with a licensed investment professional as necessary. Past performance is not indicative of future results.
Member discussion