BiQ Journal: Atrioventricular Interval Modulation (AVIM) and the Future of the Cardiac Bioelectronic Pharmacy (Part 1)
Orchestra Biomed (OBIO) is a company I have been following for some time, and I officially added it to the BiQAP this past October. At the time, shares were trading at around $2.50 and closed on Friday at $4.13.
OBIO is a small-cap medtech company developing AVIM Therapy for the treatment of uncontrolled hypertension and Virtue SAB for the treatment of Atherosclerosis. AVIM and Virtue SAB have both been granted Breakthrough Device designations by the FDA. While Virtue SAB is a very interesting and potentially high-value platform in its own right, this article focuses specifically on OBIO's AVIM Therapy platform. As is usually the case, my goal in this article is to highlight key information and provide my own perspective and insights, not to repeat information readily available via online publications. This article is also not meant to present a comprehensive investment thesis. As a starting point, I highly recommend that readers review the Orchestra Biomed website and corporate presentation for background on the company and AVIM Therapy.
AVIM stands for Atrioventricular Interval Modulation. In its simplest form, AVIM is a software-based therapy that turns a standard pacemaker into a bioelectric device for treating hypertension. In essence, AVIM uses an electrical signal to force the heart's bottom chambers to beat a split-second earlier than usual. This reduces preload by preventing the heart from filling completely. Since there is less blood to pump, this results in an immediate, predictable, and durable drop in blood pressure. Three of the key advantages of AVIM Therapy can be summarized as follows:
- "Always-on" Therapy: It treats high blood pressure 24/7 without requiring the patient to take any action, such as remembering to take a pill.
- Zero Extra Surgery: For patients who already need a pacemaker, it is a simple software "unlock" that adds no extra risk or hardware.
- Mechanical vs. Chemical: It lowers blood pressure by altering pumping mechanics. There are no systemic side effects, no DDIs, and results are immediate, durable, and highly predictable.
OBIO has entered into a worldwide licensing agreement with Medtronic to commercialize AVIM for use with standard dual-chamber transvenous pacemakers. As readers are probably aware, Medtronic is the global leader in the cardiac pacemaker market, a position it has held for decades. It is the leader in both the traditional transvenous market and the high-growth leadless segment, controlling over 50% of the worldwide market share. This gives OBIO instant global distribution of AVIM if the therapy is eventually approved. Furthermore, Medtronic also has an option to expand its partnership with OBIO to include leadless pacemakers via its market-leading Micra Transcatheter Pacing System, which represents the fastest-growing segment within the worldwide pacemaker market.
On Thursday, April 23rd, OBIO published a press release highlighting data to be presented at the Heart Rhythm Society 2026 Annual Meeting (HRS 2026) this weekend. Although more data is likely to be presented at the meeting, the results included in public releases so far were very encouraging and can be summarized as follows:
- Immediate average reduction of 13.2 mmHg in office systolic blood pressure (oSBP).
- 97% responder rate of >5 mmHg reduction in oSBP.
- 89% of patients achieved an ambulatory systolic blood pressure (aSBP) of <140 mmHg, and 58% achieved aSBP <130 mmHg at 6 months.
- Sustained reductions in aSBP out to 3.6 years in early cohorts.
- 0% MACE were reported as being related to the therapy at the 6-month primary endpoint.
- Blood pressure returned to baseline with no rebound hypertension when the device was switched off.
- Long-term use was associated with favorable reverse LV remodeling (reduced LVEDV and LVESV).
- When the device was deactivated for a "washout" period and then reactivated, the blood pressure-lowering effect immediately returned to its previous levels.
OBIO has also demonstrated that treatment benefits were immediate upon initiation of therapy, ceased when the therapy was switched off, and returned to previous levels when the therapy was switched back on. Additionally, AVIM Therapy was also found to induce reverse remodeling in older, higher-risk patients.
While the blood-pressure-lowering benefits are very encouraging, what I find even more interesting is AVIM's ability to induce reverse remodeling. Mechanistically, I think this makes sense:
- By shortening the time between the top and bottom chambers beating, AVIM prevents the left ventricle from filling up completely. This immediate reduction in preload volume means the heart muscle fibers aren't stretched as thin with every beat.
- The therapy tricks the nervous system into relaxing the body's blood vessels. This lower systemic resistance, or afterload, means the heart doesn't have to "muscle" its way through each contraction. As seen in the latest data, this reduces the "stroke work" (the heart's energy expenditure) without sacrificing the actual volume of blood pumped.
- Once you lower both the preload and the afterload, you have significantly reduced wall stress. In response to this lighter workload, the heart stops the emergency "bulking" process, or hypertrophy. Over time, the thickened muscle thins out, and the overstretched chambers return to a more compact, efficient size.
What makes this approach so exciting is that AVIM works through physics, not chemistry. Mechanically unloading the heart 100,000 times a day creates the perfect environment for reverse remodeling—potentially turning a stiff, enlarged, hypertensive heart back into a more flexible, more athletic one. There are no additional medications to take, no DDIs, no systemic adverse effects, and the therapy continues to work 24 hours a day, 7 days a week, with 100% compliance. Data in older patients highlights AVIM's ability to induce reverse remodeling, even in patients whose hearts have become too "stiff" for drugs to manage effectively. The graphic below is from an earlier data cut, as the most recent materials from HRS 2026 aren't publicly available yet. It can also be found in the company's corporate presentation.
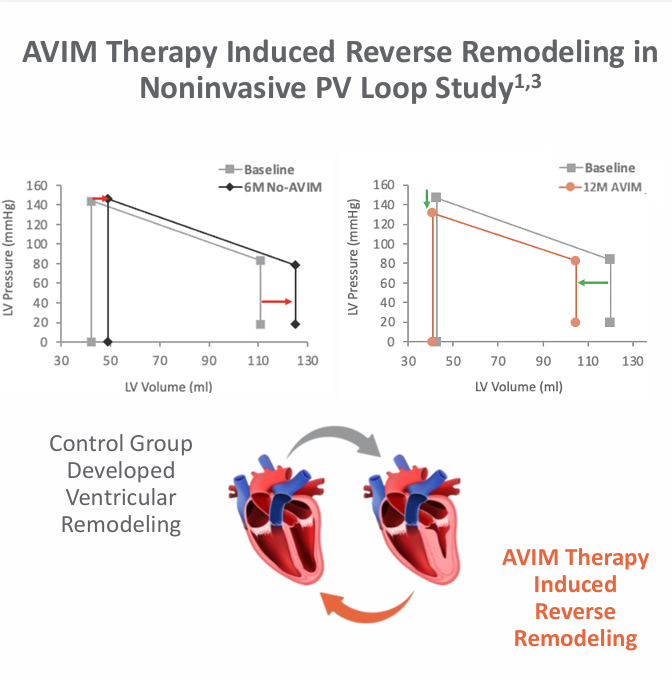
In its pivotal BACKBEAT trial, OBIO is studying the ability of AVIM Therapy to treat patients who already need a pacemaker and have uncontrolled hypertension despite standard-of-care drug therapies. So what is the revenue potential for AVIM in this subpopulation of patients?
Approximately 1.1 million dual-chamber pacemakers are implanted globally each year. This does not include leadless pacemakers, which represent the fastest-growing segment of the market; however, since the current BACKBEAT study focuses only on dual-chamber transvenous pacemakers, we will use 1.1M as the starting point for this analysis. Of this population, 70% to 80% also have hypertension, and 50% remain uncontrolled despite taking multiple medications. This creates an immediate market of roughly 400K to 500K new patients per year globally. For the purposes of this illustration, I am focusing only on newly implanted pacemakers and deliberately excluding patients with existing compatible pacemakers who would become instantly eligible to "upgrade" their devices to the new AVIM algorithms, which represents a significant additional pool of patients (a recent protocol update in 2025 allows for software retrofitting of existing compatible devices).
OBIO has provided a revenue range of $500-$1600 payable to OBIO via royalties, per AVIM activation. The range is due to the fact that the royalty is based on the pacemaker's selling price, which can vary by country. I would expect closer to $1500-$1600 in major Western markets, and more towards the lower end of the range in markets such as India or China. If we assume an average of $1000 in revenue to OBIO per AVIM activation in the subpopulation of patients identified above, and we further assume 20% market penetration of Medtronic's 50% market share, this would represent approximately $40M in annual royalty-based revenue to OBIO. (Note that in the early years, a portion of this revenue will go towards paying back royalty-based financing agreements OBIO entered into with Ligand Pharmaceuticals and Medtronic.) Although $40M may not sound like much, since it is pure high-margin royalty revenue, we can apply a 15-20x multiplier to arrive at a $700M back-of-the-napkin valuation. Again, this represents only the initial subpopulation of patients targeted in the BACKBEAT trial, includes only newly implanted devices, assumes no growth in Medtronic's market share, and excludes any contribution from other products in OBIO's pipeline.
From Medtronic's perspective, after backing out the royalty paid to OBIO, I estimate incremental revenue of around $2,000 per device on average. With roughly 500K Medtronic devices implanted annually (excluding revenue from activation on existing devices), and assuming the same 20% market penetration, this represents approximately $200M in incremental net revenue to Medtronic for activating the algorithm.
One side note: while I am assuming 20% market penetration, the actual penetration will depend on how Medtronic plans to market AVIM. AVIM will likely be preinstalled on every pacemaker Medtronic sells. What's currently not known is whether Medtronic will add the cost of the AVIM "upgrade" to the initial cost of the hardware, thereby front-loading the revenue, or charge for the AVIM algorithms only once they are activated. For purposes of this illustration, and to be conservative, I am assuming the latter scenario. However, it's equally possible that we could see a mix of the first and second scenarios, depending on the end-market. Any "pre-selling" of AVIM would represent additional upside from my estimates.
I consider these to be very conservative estimates based on the assumptions I've used above. However, I think BACKBEAT only scratches the surface of OBIO & Medtronic's strategic vision of AVIM's potential. This is where the concept of what I'm terming the "Cardiac Bioelectronic Pharmacy" comes into play. I will explore this further in my next article, which will be available to BiQ Premium members and posted to my website in the coming days.
OBIO share price at time of publication: $4.13
BiQ ACB: $2.48
Not a BiQ Premium member, or have a friend who may be interested? Try BiQ Premium for $50 per month, or $375 (25% off) for the first year.
Biotech iQ is 100% subscriber supported. If you find this information helpful, please spread the word. You can also follow me on X @ Biotech iQ (_Biotech_iQ).
Biotech iQ is not an investment professional, and nothing on this page or this website should be considered investment advice. Please consult with a licensed investment professional as necessary. Past performance is not indicative of future results.
Member discussion